
(b) The aldehyde, carboxylic acid and ester will be reduced to the same product, benzyl alcohol. Since the -OR group is a stronger electron donor (resonance) than the alkyl group of the ketone, the ester is less reactive than the ketone. Under the reaction condition s the carboxylic acid will deprotonate to give the carboxylate which is a very poor electrophile (after all, it has a negative charge !) so the ester is more reactive than the acid. Because of their enhanced acidity, carboxylic acids react with bases to form ionic salts, as shown in the following equations. 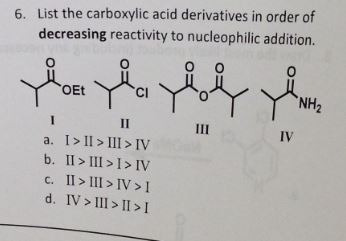
Aldehydes are more reactive than ketones !!! as they are less hindered and the alkyl group in the ketone is a weak electron donor. Arrange the following compounds in order of increasing acidity and. EXPLAINATION: Electron withdrawing groups like -NO2, increases the acidity. TUTORIAL 17 : CARBOXYLIC ACID AND ITS DERIVATIVES 17.1 Introduction to carboxylic. Consider the electrophilicity of the carbonyl group in each compound in each pair. The decreasing order of their acidic strength: NO2CH2COOH > FCH2COOH > C6H5COOH. CH3CH2OH, CH3COOH, ClCH2COOH, FCH2COOH, C6H5CH2COOH. Arrange the following in decreasing order of their acidic strength and give the reason for your answer. Overall the products formed in this reaction are 2-Methylpropanoic acid, 3-Methylbutanoic acid, ethanoic acid and methanoic acid. The aldehyde and ketone will undergo nucleophilic addition, the acid and the ester nucleophilic acyl substitution. Ketone is then further oxidized to carboxylic acids. : First task should be to identify the functional groups: carboxylic acid, ketone, aldehyde, ester. 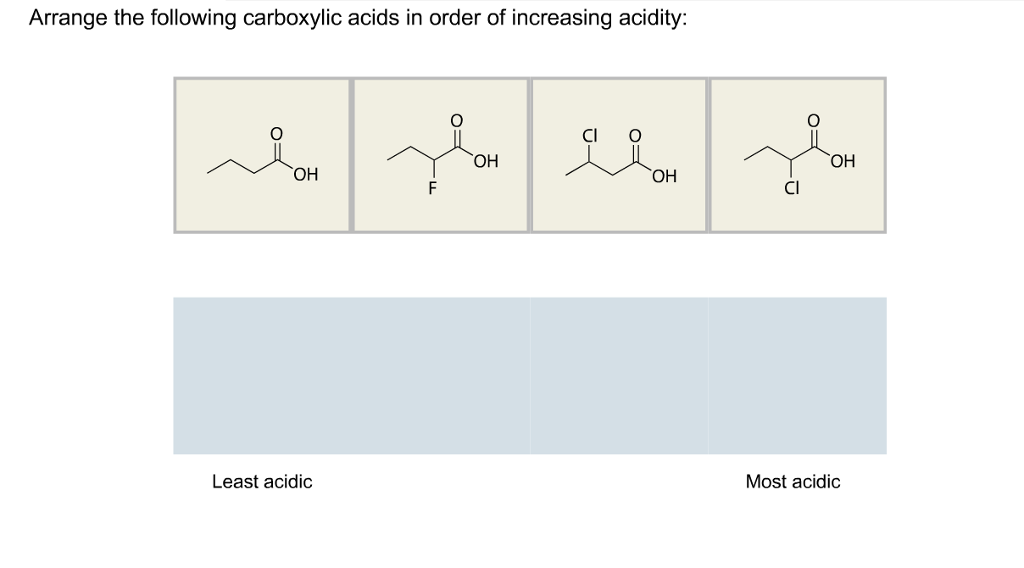
(b) 2 moles CH3CO2H (d) CH3CO2H and HN(CH3)2Ħ LiAlH4 is a source of H- (a nucleophile) which functions as a reducing agent. Arrange the following carboxylic acids in order of acidity.ClCOOHCOOHCOOHCOOHThe strongest acid is The second strongest is The third strongest is The weakest. 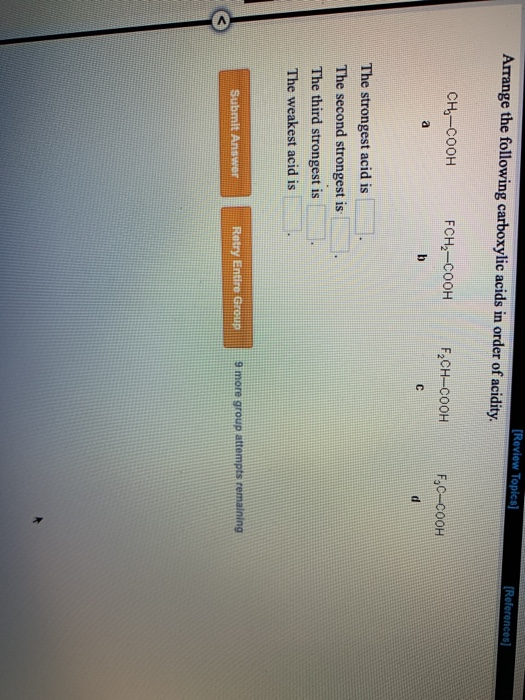
Hydrogen and metals combined in an alkaline solution produce. (b) LiAlH4 / THF then acidic work-up (d) NaOHģ.-How could you use 1-bromobutane to prepare each of the following carboxylic acids ? (a) propanoic acid (b) butanoic acid (c) pentanoic acid (d) hexanoic acidĬarboxylic Acid Answers 1: (i) increasing acidity : CH3CH2OH > acid anhydride ester amide chloride 5 All the carboxylic acids in Qu 1 are derivatives of ethanoic acid, so they all : give the same carboxylic acid. Carbaryl acid is the name given to organic compounds that contain carboxylic acid groups. The carboxylic acids are acidic in nature because the hydrogen in -COOH group can be given forming carboxylate ion. Carboxylic Acid Questions 1.-Arrange the following in order of (i) increasing acidity, (ii) increasing pKa : (a) CH3CO2HĢ.-What would be the major products of the reactions of (i) butanoic acid and (ii) benzoic acid with each of the following: (a) SOCl2, Et3N (c) (CH3)2CHOH / H+ / heat The acidity of carboxylic acids is higher in comparison to simple phenols as they react with weak bases like carbonates and bicarbonates to liberate carbon dioxide gas. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
